The Best Help in Services & ai medical
Transthyretin (ATTR) cardiac amyloidosis is a progressive infiltrative cardiomyopathy caused by the deposition of misfolded transthyretin (TTR) protein within the myocardium. Effective treatment requires a dual strategy:
01
Disease-modifying therapies
targeting the underlying TTR protein cascade
02
Supportive cardiac management
to control heart failure and conduction complications
Early diagnosis is critical, as disease-modifying therapies are most effective before advanced structural damage occurs.
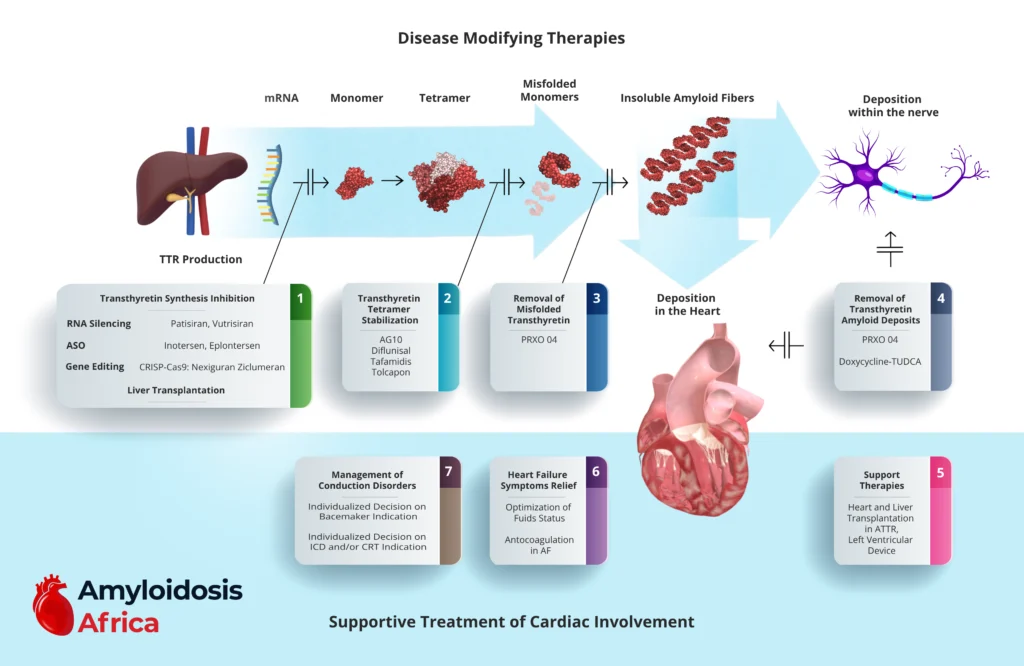
Understanding the Therapeutic Targets
ATTR cardiomyopathy develops through a multistep biological cascade:
1
TTR gene transcription
2
TTR monomer production
3
Tetramer dissociation
4
Misfolded monomers
5
Amyloid fibril formation
6
Tissue deposition in the heart and nerves
Modern therapies intervene at different stages of this pathway.
1. Inhibition of TTR Production (Gene Silencing & Editing)
These therapies reduce hepatic production of transthyretin, lowering circulating TTR levels and slowing amyloid formation.
RNA Interference (RNAi)
Patisiran
Vutrisiran
These small interfering RNA therapies target TTR mRNA in hepatocytes, significantly reducing serum TTR levels.
Antisense Oligonucleotides (ASO)
Inotersen
Eplontersen
These inhibit TTR mRNA translation, reducing protein synthesis.
Gene Editing
CRISPR-Cas9 (e.g., Nexiguran Ziclumeran)
Emerging therapies aim to permanently silence the TTR gene through in vivo genome editing.
Liver Transplantation
Historically used in hereditary ATTR (hATTR), liver transplantation removes the primary source of mutant TTR production. However, it is now less common due to targeted pharmacologic therapies.
2. Transthyretin Tetramer Stabilization
Tetramer dissociation is the rate-limiting step in amyloid formation. Stabilizers bind TTR and prevent dissociation into misfolded monomers.
Approved and Investigational Stabilizers
Tafamidis (standard of care in ATTR-CM)
Diflunisal
Acoramidis (AG10)
Tolcapone
Tafamidis has demonstrated mortality and hospitalization benefits in ATTR-CM and is currently the cornerstone disease-modifying therapy.
3. Removal of Misfolded Transthyretin
Emerging therapies aim to enhance the clearance of circulating misfolded TTR species.
PRX004 (investigational monoclonal antibody)
These therapies seek to neutralize toxic TTR aggregates before tissue deposition.
4. Removal of Amyloid Deposits
Therapies targeting established amyloid fibrils aim to reverse organ dysfunction.
PRX004 (dual mechanism potential)
Doxycycline + TUDCA (investigational approach)
Amyloid removal remains an area of active research and represents a promising future direction.
Supportive Treatment of Cardiac Involvement
Even with disease-modifying therapy, most patients require careful cardiac management.
5. Heart Failure Management
Heart failure in ATTR-CM is predominantly restrictive with preserved or mildly reduced ejection fraction.
Key Principles:
Careful fluid management
Loop diuretics as first-line therapy
Mineralocorticoid receptor antagonists when tolerated
Avoid excessive preload reduction
Caution:
ACE inhibitors, ARBs, and beta-blockers are often poorly tolerated due to low stroke volume.
Calcium channel blockers and digoxin should generally be avoided due to potential toxicity in amyloid hearts.
Anticoagulation
Atrial fibrillation is common and carries a high thromboembolic risk. Anticoagulation is often recommended regardless of CHA₂DS₂-VASc score.
6. Conduction System Disease Management
ATTR frequently affects the conduction system.
Management includes:
Pacemaker implantation for symptomatic bradycardia or AV block
Individualized decision-making for ICD placement
Consideration of CRT in selected patients
Electrophysiologic monitoring is essential due to progressive conduction abnormalities.
7. Advanced Supportive Therapies
Heart Transplantation
May be considered in carefully selected patients, particularly younger individuals with hereditary disease.
Combined Heart–Liver Transplantation
Rare but considered in selected hereditary cases.
Mechanical Circulatory Support
Left ventricular assist devices (LVAD) have limited use due to restrictive physiology but may be considered in specific contexts.

Personalized and Multidisciplinary Care
Management of ATTR cardiac amyloidosis requires coordination between:
Cardiologists
Heart failure specialists
Neurologists
Geneticists
Electrophysiologists
Multidisciplinary amyloidosis clinics
Genetic testing is mandatory in all confirmed ATTR cases to distinguish:
Hereditary ATTR (hATTR)
Wild-type ATTR (wtATTR)
This distinction guides family screening and therapeutic strategy.
Treatment in the African Context
In Africa, treatment access remains unequal. Key priorities include:
Expanding diagnostic capacity
Improving access to disease-modifying therapies
Strengthening clinician education
Developing Africa-led registries and outcome data
Integrating ATTR into heart failure pathways
Early recognition of red flags, bilateral carpal tunnel syndrome, unexplained LV hypertrophy, apical sparing pattern on strain imaging, neuropathy, and low voltage ECG, can significantly improve outcomes.
Future Directions in ATTR Therapy
The therapeutic landscape is evolving rapidly:
Long-acting RNA therapies
Gene editing approaches
Amyloid-clearing antibodies
Combination therapy strategies
The future of ATTR treatment lies in earlier diagnosis, combination disease-modifying strategies, and equitable global access.
Key Takeaways
- ATTR cardiac amyloidosis is treatable, particularly when diagnosed early.
- Tafamidis remains the cornerstone stabilizer therapy.
- Gene-silencing therapies significantly reduce TTR production.
- Supportive heart failure care remains essential.
- Multidisciplinary management improves outcomes.
- Africa must expand diagnostic and therapeutic access to reduce late-stage presentation.

We advance cardiac amyloidosis care across Africa through collaboration, training, and research. Join us to change the future of heart health and empower local clinicians with knowledge and tools.
- Support cardiac care in Africa
- Give what you can to help
- Join our research network!
- Follow updates and news



